We all want new and better medicines. Drugs that provide incremental benefits (often called “me too” drugs) are good, but truly breakthrough therapies are even better. However, how can you tell how “new” a treatment is? Is there a way to quantify this?
In fact there is. One way to do this is based on the molecular structure of the treatment. For example, him tanimoto distance It measures the fraction of chemical characteristics that the two chemical compounds share. Nikolova et al. 2004 summarizes this approach which is calculated as follows:
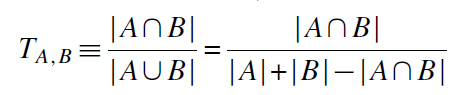
Krieger, Li and Pap smear 2022 Use this method to quantify how new drug development has much more positive informational spillover effects on drug development than incremental drugs. This article provides an example of an application of the Tanimoto distance below.
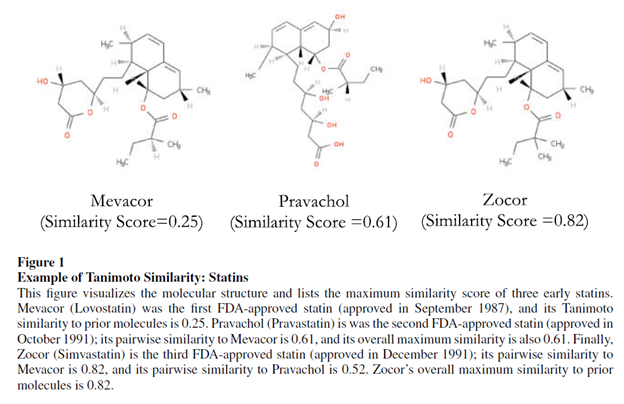
Using the Tanimoto distance as a measure of novelty, the authors find the following:
…new drug candidates are less likely to win FDA approval, but are based on more valuable patents. Consistent with a simple model of costly external financing, we show that a positive shock to firms’ net worth leads firms to develop more novel drugs. This suggests that even large companies can behave as if they are risk-averse, reducing their willingness to invest in potentially valuable radical innovations.
You can read the full article. here.








